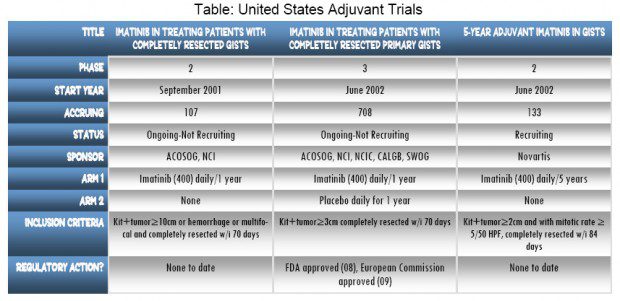
Adjuvant trials don’t always get the same attention as second and third line trials for advanced GIST patients. They can also take up to seven years to get results. But when they do get reported the news can be earth moving. In late 2008, the Food & Drug Administration approved imatinib for adjuvant use in patients whose GIST has been completely removed. The European Medicines Agency (EMEA) followed with a recommendation for approval in March 2009, but the phase III trial that produced the results supporting those decisions started in June 2002. It was halted early in April 2007 because a clear advantage was seen in recurrence-free survival for patients in the active imatinib arm. Patients on imatinib had less than half the risk of recurrence compared to patients on placebo. That trial had been studying imatinib versus placebo for one year after surgery with over 700 patients participating in the United States. Although the results were significant, the question remains about what to do after that first year. How long should patients stay on imatinib?
In a parallel phase II adjuvant trial of imatinib alone in high-risk GIST patients (tumors greater than 10 cm) researchers found a similar response pattern over the one year of the trial treatment period. They also found that when adjuvant treatment was stopped there was a significant increase in the rate of recurrence beginning six months after treatment was discontinued.
What’s new?
Novartis has initiated a new adjuvant phase II trial: imatinib in patients who have had complete surgical removal of their GIST. Novartis plans to recruit 130 patients at over 30 sites in the United States for a five-year study of imatinib without a placebo comparison. The trial started recruiting in July, and twelve sites are currently open. Patients will receive 400 mg of imatinib daily and followed for the first five years of the study. At the end of treatment patients will be followed for another five years. Plans are to recruit patients up to December 2010.
Who should consider
Patients who have had tumors larger than two cm and with mitotic counts equal to or greater than five per 50 High Power Fields (HPF) will be eligible. Non-gastric tumors must be greater than five cm. Mitotic Index criteria for nongastric are not given but appear to include both less than five and greater than five mitoses per HPF. By definition these patients are at significant risk for recurrence. Patients must have a histological diagnosis of primary GIST. Tumors removed must test positive for KIT (CD117). All tumors must have been completely removed at the time of surgery (i.e. no metastatic disease on post surgery scans). Patients must enter the trial within 12 weeks of primary tumor surgery. Prior use of imatinib is restricted, and other prior therapies are excluded. There are additional criteria in the detailed PubMed listing under NCT00867113. Novartis also has a new online form to see if you qualify at www.novartisclinicaltrials.com. Select GIST from the conditions list and then select this trial.
Questions to ask
This description for trial does not specify whether patients will receive primary mutational analysis as part of the trial. It would be advisable to have mutation analysis done as part of the initial diagnosis process before considering this trial. Patients who are wild type, exon 9, or PDGFRA mutation D842V do not always get optimal response to imatinib 400 mg daily. As always, you should consult your oncologist when considering a clinical trial.