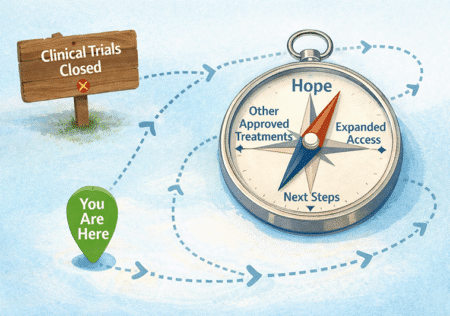
 Pharmaceutical companies may create an expanded access program (sometimes called compassionate use) when a clinical trial is no longer enrolling but the medicine still shows promise. This program gives patients with serious or life-threatening illness another way to try the investigational treatment if they cannot join the trial and have few or no other options. It allows doctors to request the drug for their patients while the company continues to study it and work toward possible approval. Expanded access can also help researchers learn more about the drug’s safety in real-world use, while offering hope to patients who need more choices.
Pharmaceutical companies may create an expanded access program (sometimes called compassionate use) when a clinical trial is no longer enrolling but the medicine still shows promise. This program gives patients with serious or life-threatening illness another way to try the investigational treatment if they cannot join the trial and have few or no other options. It allows doctors to request the drug for their patients while the company continues to study it and work toward possible approval. Expanded access can also help researchers learn more about the drug’s safety in real-world use, while offering hope to patients who need more choices.
The quick FAQs:
What is Expanded Access?
Expanded Access Programs (EAP), also known as compassionate use, allow patients to receive investigational clinical trial drugs outside of clinical trials settings. This option may be available for patients who have exhausted all approved treatment alternatives.
Am I eligible?
Each EAP has its own eligibility criteria. In general, if you do not have other approved treatment options, an expanded access program may be an option for you.
How does it work?
EAPs are administered by pharmaceutical companies. Approval is granted at the company’s discretion based on program requirements. Your doctor will submit a request on your behalf to the program to see if you may qualify.
Are there limitations?
Participation in some expanded access programs may be limited, as some hospitals may face restrictions on the number of clinical trial related programs they can conduct.
If you are considering starting a new treatment, reach out to us. We may be able to help you understand what options or programs could be available to you.
Interested in learning more?
If you are interested in current expanded access programs, please contact:
ssingh@liferaftgroup.org or (973)-980-2945

