Note that this report is based on an abstract submitted to the 2011 European Multidisciplinary Cancer Congress. As such, it should be considered as preliminary.
This post provides details of the trial results referred to in this post: LRG Survey Seeks to Understand Differences in Sutent Dosage Schedules
2011 European Multidisciplinary Cancer Congress –
Abstract 9421 – Optimizing Management of Sunitinib Treatment in a Worldwide Treatment-use Trial of Patients with Advanced Gastrointestinal Stromal Tumors (GIST)
P. Reichardt, Y.K. Kang, P. Rutkowski, J. Schuette, L.S. Rosen, B. Seddon, S. Yalcin, L. Chen, K. Fly, G.D. Demetri
- This was a treatment use trial that provided Sutent access to 1124 GIST patients after the end of the phase III trial and prior to approval of Sutent (Feb, 2006 in the USA) (NCT00094029).
- The intended dose/schedule for all patients was 50 mg/day for 4 weeks followed by 2 weeks of no drug. This schedule was repeated as long as patients benefited.
- Dose/schedule changes were permitted. Patients that changed dose or schedule are reported as Flexible dose (FD).
- SSDS – Strict starting dosing schedule, strict adherence to intended dose/schedule
- 599 patients
- FD – Flexible dose, changes were made to the dose or schedule
- 525 patients
- Baseline characteristics were generally well balanced between the two groups except:
- Flexible dose – had more women and more Asians
- 46% women
- 23% Asians
- Perhaps indicating that these two groups did not tolerate Sutent as well
- Flexible dose – had more women and more Asians
- Strict dose
- 35% women
- 13% Asians
|
Treatment Cycles (started) |
Treatment interruptions |
Time to tumor progression (median) |
Overall Survival |
|
| Flexible dose (599 pts) |
9 |
73% (5% of days on treatment) |
12.6 months |
23.4 months |
| Strict dose (525 pts) |
3 |
39% (7% of days on treatment) |
5.2 months |
11.1 months |
| Adverse events were more common in the Flexible dose group. It seems likely that many of these patients were placed on flexible dosing because they were not tolerating the strict dose as well. | ||||
Authors conclusions: Patients with GIST who received sunitinib using a flexible dose approach to manage adverse events remained on treatment longer than those who received the strict dose/schedule and exhibited better clinical outcomes (longer time to tumor progression and longer overall survival).
Treatment guidelines as of 8/2/2012:
- FDA prescribing information for Sutent (PDF)
- GIST and RCC – 50 mg/day, 4 weeks on treatment followed by 2 weeks off
- Pancreatic neuroendocrine tumors (pNET), 37.5 mg/day continuously (no scheduled days off)
- NCCN guidelines for GIST (USA) (link to guidelines page (under Sarcoma), access requires registration)
- 50 mg/day, 4 weeks on, 2 weeks off
OR
- 37.5 mg/day continuous
- ESMO guidelines for GIST (Europe, PDF)
- 50 mg/day, 4 weeks on, 2 weeks off
- 37. Mg/day continuous can be considered an option on an individualized basis.
LRG Commentary
It is interesting to note that patients that received a flexible dose/schedule did twice as well as patients that remained on the strict 4 week on/2 week off schedule at 50 mg/day. This was true both for time to tumor progression and for overall survival. This was not a randomized trial however. A previous report in kidney cancer patients found that severe toxicities were much more common in females and older patients. This may explain why there were more females in the flexible dose; they may have required a dose reduction/change in schedule because of higher toxicity. Female gender has been noted in many reports to be a favorable factor in GIST (women tend to do better than men). So it’s possible that having more women in the flexible dose group may have increased survival. The magnitude of the difference is hard to ignore however. It would be interesting to see a sub-analysis of the data comparing men in both groups (FD to SDSS) to each other and comparing women in both groups to each other.
The large benefit observed warrants further investigation. In the interim, both the NCCN guidelines (USA) and the ESMO guidelines (Europe), permit either dose/schedule.
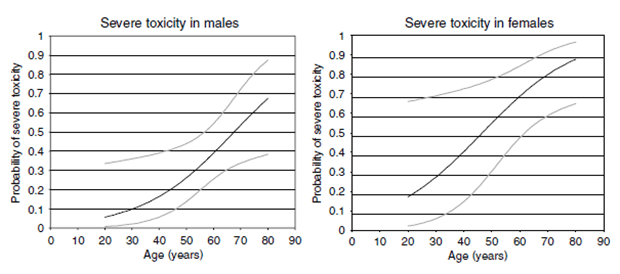
Figure 1 – Probability of severe toxicity from sunitinib (50 mg per day 4 weeks on and 2 weeks off) in patients with advanced RCC based on the following model: Probability of severe toxicity in male patients=exp (−3.986+0.059*age)/(exp (−3.986+0.059*age)+1) Probability of severe toxicity in female patients=exp (−2.750+0.059*age)/(exp (−2.750+0.059*age)+1) Grey lines represent confidence intervals.
van der Veldt AAM, Boven E, Helgason HH, van Wouwe M, Berkhof J, de Gast G, Mallo H, Tillier CN, van den Eertwegh AJM, Haanen JBAG: Predictive factors for severe toxicity of sunitinib in unselected patients with advanced renal cell cancer. Br J Cancer 2008, 99:259–265.


