With the advent of Imatinib, tremendous progress has been made in the treatment of patients with GIST.
Imatinib is a drug that can be taken orally that inhibits the function of KIT, a protein that is expressed in GIST cells. In these cells, the KIT protein undergoes a mutation (change) in the DNA that causes it to send an activating signal to the GIST cells and induces them to grow.
With Imatinib’s ability to inhibit the KIT protein, we have seen a dramatic increase in the life expectancy of patients with this tumor. Unfortunately, after a period of treatment with Imatinib, other mutations can eventually occur in the KIT molecule, rendering it insensitive to Imatinib. Therefore, a lot of research in past years has focused on the discovery and testing of novel inhibitors that can inhibit KIT proteins that have undergone these secondary mutations.
The Immune System and Cancer
I would like to give a brief description of a different approach to cancer that involves the immune system. Many researchers have been working in this approach for decades, but only in recent years have very promising results been obtained in clinical trials; trials that have so far focused on tumors other than GIST. In this approach, different parts of the immune system are used to battle tumors.
The immune system consists of a highly complex collection of different cell types. Each of these cell types can have different or partially overlapping functions with other cell types.
Some examples of the most common cell types in the immune system are T-lymphocytes, macrophages, and B-lymphocytes. T-lymphocytes can have many different functions, including killing normal human cells that have been infected by a virus. Macrophages are generic cleaners that can eat up cells recognized as foreign or that are otherwise labeled as abnormal in a variety of manners. B-lymphocytes make immunoglobulins, which are proteins that can specifically bind to a tumor cell and cause it to be eaten up (phagocytosed) by a macrophage.
Helping T-lymphocytes Kill Tumor Cells
In past years, a series of antibodies have been developed that can increase the ability of the human immune system, specifically T-lymphocytes, to recognize tumor cells as “foreign” so that they then can be destroyed.
At the recent ASCO meeting, the combination of two of these antibodies, one directed against CTLA-4 and the other against PD-1, showed a response rate of almost 50% in melanoma patients. Although the trial was small, this response rate was very high, and the hope is that this work will continue to provide good results and that this approach will also be tested in patients with GIST. A number of publications describe this approach (1-5).
Helping Macrophages Attack Tumor Cells
Macrophages are cells that clean up debris or destroy cells that are recognized as “foreign”. A second approach in immunotherapy is in development in an earlier stage, but hopefully will go to a clinical trial in the next year. This approach relies on the ability of certain antibodies to “uncloak” tumor cells that are hiding from macrophages.
Tumor cells can protect themselves from being seen as foreign and thus from being “phagocytosed” or eaten up by macrophages by expressing a protein called CD47 on their surface. Antibodies against CD47 protein can interfere with this camouflage effect and can allow phagocytosis to proceed (See Figure 1).
This approach has been quite successful in experiments where human tumors were grown as subcutaneous nodules in mice. While it remains to be seen whether these results can be translated to the human setting, the findings are quite exciting.
In addition to the range of cancers that were examined by the Weissman laboratory, we collaborated in a study to look at the treatment of leiomyosarcoma tumors in this manner. We showed that antibodies directed against CD47 could inhibit the growth of human leiomyosarcoma (LMS) cells in mice. The LMS cell lines were obtained from the laboratory of Dr. Jonathan Fletcher. The mice receiving the anti-CD47 antibodies did much better in that their tumors became smaller and they had many fewer lung metastases (6).
These experiments still need to be performed with GIST cells, but if successful, they could ultimately lead to a novel therapeutic approach in patients with GIST.
Recently, we collaborated in a study to show that in addition to antibodies against CD47, one could also inhibit the function of CD47 by a synthetic protein that was generated to bind the CD47 protein at a very high level of strength. This so-called “synthetic variant” of a protein called SIRPa was also highly successful in animal experiments and in vitro experiments in a range of tumors, and hopefully we will extend these studies to include GIST cells in the future (7).
So far, these studies have been performed in carcinomas, lymphomas, and leiomyosarcoma. So, we hope to start similar experiments using GIST cell lines.
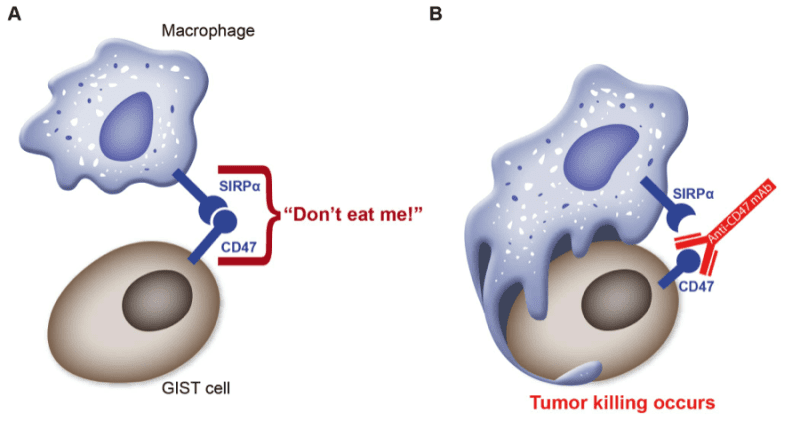
Figure 1: A protein called CD47 on the surface of GIST cells binds to SIRPa, a protein on the surface of macrophages, this send a powerful “don’t eat me” signal to the macrophages (panel A). A therapeutic antibody against CD47 disrupts the binding of CD47 to SIRPa; the “don’t eat me” signal is not delivered to the macrophage and tumor killing occurs
Using Antibodies in Immunotherapy, ROR2 As a Candidate Target
A third approach in immunotherapy relies on the use of antibodies. Antibodies are proteins that are synthesized by B-lymphocytes, a cell type specialized in generating these proteins. B-lymphocytes can generate immunoglobulins in such a way that they only react with one specific protein, like one key may fit only one specific lock. As a result, an antibody against the KIT protein will react only with KIT and not with the other thousands of other proteins in the body, for example.
Antibody therapy has become widely used in the past decades, and some of the currently best-selling drugs are antibodies. Some examples are Rituximab, an antibody that is used in treating leukemias and lymphomas and Trastuzumab, an antibody that is used to treat patients with breast cancer.
For antibody treatment to be successful, it is useful if the protein targeted by the antibody is expressed at low levels in normal human tissues but is expressed at fairly high levels in tumor samples.
One such protein is called ROR2, which is an example of a protein that is found in very few adult human tissues, but is expressed at high levels in some (but not all) GIST and LMS tumors. Just like the KIT protein, the ROR2 protein is a member of the family of receptor tyrosine kinases (RTK), molecules that are present on the surface of the cells. The RTKs can be activated when a specific protein called a ligand binds to them on the cell surface, resulting in an activating signal that is then transmitted to the cells. For ROR2, the ligand is Wnt5A (See Figure 2).
The significance of ROR2 is supported by the fact that we found that tumors that expressed this marker behaved more aggressively than tumors that did not (8).
We are currently developing a monoclonal antibody that can target the ROR2 protein. This antibody will initially be studied in cell culture for its effect on GIST cell growth. If successful, the study will be performed in mice bearing GIST tumors. Finally, if these are successful, we hope to develop an ROR2-based clinical trial for GIST patients.
The development of these reagents is unfortunately a very slow process and it will be several years before we will know how successful this approach can be.
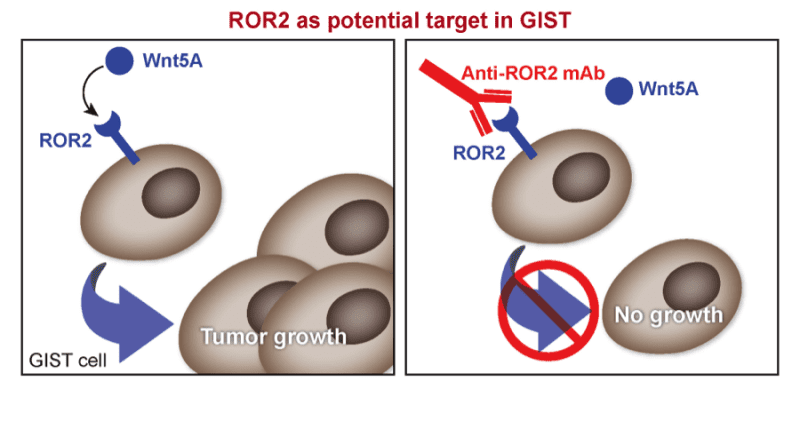
Figure 2: Activation of ROR2 occurs when Wnt5A binds to it, this causes cells to grow. An antibody against ROR2 could prevent the binding of Wnt5A and stop tumor growth.
Using Antibodies in Immunotherapy, the KIT Protein As a Target
While the ROR2 protein is expressed on only about 30% of GIST tumors, there is of course one protein that is expressed on almost all GIST tumors: the KIT protein.
A mutation in the gene coding for the KIT protein is what causes the GIST tumors to grow. While KIT can be successfully inhibited with small molecule inhibitors such as Imatinib, the KIT protein itself also forms a potential target for immunotherapy with antibodies.
We believe that we may have found strong support for this approach (9). Using cell lines made of GIST tumors that we received from Dr. Jonathan Fletcher’s lab, we could show that adding an anti-KIT antibody called “SR1” to cultures of these cell lines would decrease their growth rate. In addition, when we isolated macrophages from a mouse, we could show that addition of this antibody to GIST cells “tagged” the cells in such a way that they became more easily phagocytosed by the macrophages. Finally, when we grew the GIST cell lines in the abdominal cavity of mice, we found that treatment of these mice with the SR1 antibody resulted in much smaller tumors than when a control antibody was used (See Figure 3).
While the results are very inspiring, many issues need to be resolved. First, the antibody we used was made in a mouse and this cannot be used a therapeutic drug in humans. The human body will recognize the protein from the mouse as foreign and will immediately destroy it before it can start its therapeutic effect.
A solution to this problem is to obtain an antibody that has been “humanized.” What this means is that all the fragments of the antibody that are specifically for a mouse and that will be recognized by the human body as foreign will be replaced by protein material that is found in humans.
Another concern for this therapy is that the KIT protein is expressed on an important subset of cells in the bone marrow. This cell is called the “hematopoietic stem cell” and it is the cell from which all blood elements are derived. Clearly, one would not want to destroy that cell compartment. Many experiments will be needed to see whether this negative side effect can be avoided or to which extend bone marrow transplantation given after treatment with anti-KIT antibody can restore the blood forming cells.
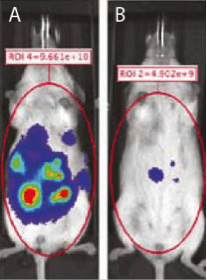
Figure 3: Mice were injected with GIST tumor cells into the abdominal cavity. Mice treated with control antibody grew large tumors (panel A) while the tumor were much smaller in those receiving anti-KIT monoclonal therapy (panel B).
Summary
In addition to the examples given above for immunotherapeutic approaches, many other approaches are being pursued by research laboratories around the world. Many of those approaches focus on tumors other than GIST, but there is a definite possibility that findings obtained in other tumor types can be applied to GIST care as well.
The few immunotherapeutic approaches described above hopefully will give some insight into novel developments in immunotherapy and show a possible approach to GIST treatment that in the future could be used in addition to the treatment by small molecule inhibitors such as Imatinib.
References
1. Mellman I, Coukos G, Dranoff G. Cancer immunotherapy comes of age. Nature. Nature Publishing Group. 2011 Dec 21;480(7378):480–9.
2. Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. Nature Publishing Group. 2012 Mar 22;12(4):252–64.
3. Topalian SL, Weiner GJ, Pardoll DM. Cancer Immunotherapy Comes of Age. Journal of Clinical Oncology. 2011 Dec 16;29(36):4828–36.
4. Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, Powderly JD, Carvajal RD, Sosman JA, Atkins MB, Leming PD, Spigel DR, Antonia SJ, Horn L, Drake CG, Pardoll DM, Chen L, Sharfman WH, Anders RA, Taube JM, McMiller TL, Xu H, Korman AJ, Jure-Kunkel M, Agrawal S, McDonald D, Kollia GD, Gupta A, Wigginton JM, Sznol M. Safety, Activity, and Immune Correlates of Anti–PD-1 Antibody in Cancer. N Engl J Med. 2012 Jun 28;366(26):2443–54.
5. Brahmer JR, Tykodi SS, Chow LQM, Hwu W-J, Topalian SL, Hwu P, Drake CG, Camacho LH, Kauh J, Odunsi K, Pitot HC, Hamid O, Bhatia S, Martins R, Eaton K, Chen S, Salay TM, Alaparthy S, Grosso JF, Korman AJ, Parker SM, Agrawal S, Goldberg SM, Pardoll DM, Gupta A, Wigginton JM. Safety and Activity of Anti–PD-L1 Antibody in Patients with Advanced Cancer. N Engl J Med. 2012 Jun 28;366(26):2455–65.
6. Edris B, Weiskopf K, Volkmer AK, Volkmer JP, Willingham SB, Contreras-Trujillo H, Liu J, Majeti R, West RB, Fletcher JA, Beck AH, Weissman IL, van de Rijn M. Antibody therapy targeting the CD47 protein is effective in a model of aggressive metastatic leiomyosarcoma. Proc. Natl. Acad. Sci. U.S.A. 2012 Apr 24;109(17):6656–61.
7. Weiskopf K, Ring AM, Ho CCM, Volkmer J-P, Levin AM, Volkmer AK, Ozkan E, Fernhoff NB, van de Rijn M, Weissman IL, Garcia KC. Engineered SIRPα variants as immunotherapeutic adjuvants to anticancer antibodies. Science. 2013 Jul 5;341(6141):88–91.
8. Edris B, Espinosa I, Mühlenberg T, Mikels A, Lee C-H, Steigen SE, Zhu S, Montgomery KD, Lazar AJF, Lev D, Fletcher JA, Beck AH, West RB, Nusse R, van de Rijn M. ROR2 is a novel prognostic biomarker and a potential therapeutic target in leiomyosarcoma and gastrointestinal stromal tumour. J. Pathol. 2012 Jun;227(2):223–33.
9. Edris B, Willingham SB, Weiskopf K, Volkmer AK, Volkmer JP, Muhlenberg T, Montgomery KD, Contreras-Trujillo H, Czechowicz A, Fletcher JA, West RB, Weissman IL, van de Rijn M. Anti-KIT monoclonal antibody inhibits imatinib-resistant gastrointestinal stromal tumor growth. Proc. Natl. Acad. Sci. U.S.A. 2013 Feb 26;110(9):3501–6.
