This year, the Life Raft Group joined the Clinical Trials Transformation Initiative (CTTI) to help make clinical trials more efficient and productive and to help accelerate new treatments. CTTI is a consortium that brings together more than 75 stakeholders from government, regulatory agencies, industry (pharma, CROs) medical institutions, academia and patient groups to identify real-world solutions to tough problems that are slowing down the development of new treatments across all diseases. Members include Celgene, Pfizer, Genentech, GSK, Merck, NIH, CDC, FDA, NORD, Dana- Farber Cancer Institute, and many others.
Over the years, the complexity and cost of clinical trials has resulted in a system that is unsustainable and often fails to provide patients with reliable and timely new treatments. CTTI’s recommendations help to inform both policy and decision-making in the interest of public health, as well as streamline the operation of clinical trials. By participating in this collaborative environment the Life Raft Group helps keep the GIST community at the forefront of innovation and the patient as a force of constructive change.
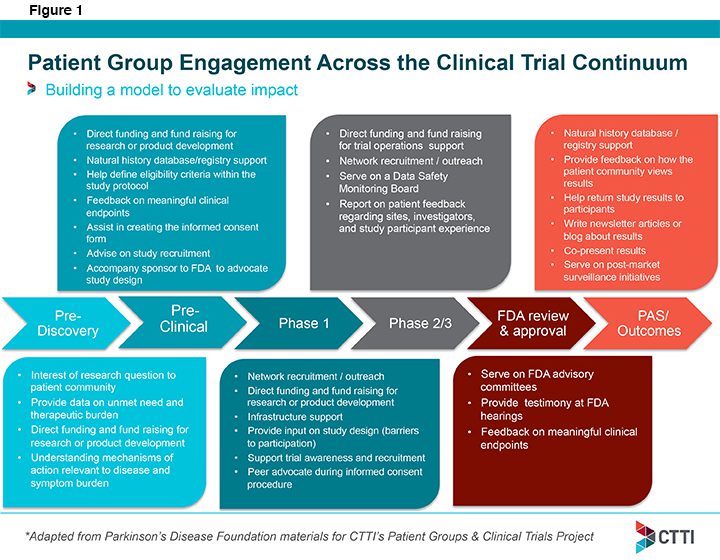
In January, Acting Deputy Executive Director Marisa Bolognese participated in CTTI’s Patient Groups and Clinical Trials summit to help determine best practices for effective patient engagement in all phases of drug development. Ultimately, the project will produce an actionable toolkit so that patient groups and sponsors can more effectively work together to create more efficient and quality-driven clinical trials. As a first step roles for patients across the clinical trial continuum were identified (see Figure 1 – below).
In March, Executive Director Norman Scherzer participated in the bi-annual steering committee meeting in Washington D.C. where new recommendations for the use of single Institutional Review Boards (IRBs) in multi-site trials were approved. The LRG has long been a strong advocate for this as repetitive IRB processes have contributed to the inefficiency and delay of clinical trials including many for GIST.
The LRG is also now involved with CTTI’s Mobile Clinical Trials project, which is looking at novel technologies that will permit patients to enroll in trials without having to travel to a central location. Such technology is already utilized for the Life Raft Group/National Institute of Health (LRG/NIH) Collaborative Tumor Boards and hopefully will continue to play an important role as we plan for innovative GIST clinical trials in the near future.

