From June 15 through June 17, 2011, the National Institutes of Health (NIH) hosted the seventh annual Pediatric and wildtype GIST Clinic. Ten patients from across the United States and Canada attended, accompanied by family members or friends. In total, 80 patients have participated in the NIH Pediatric GIST Clinic, 75 of whom have wildtype GIST. Thirty-five percent of patients have pediatric GIST, currently defined as diagnosis before the age of 19.
The major topics addressed during the clinic included the interpretation of survival data stratified by clinical parameters, the implications of Succinate Dehydrogenase subunit B (SDHB) immunostaining, and how all of this information could help refine the current definition of pediatric GIST.
The average age of patients at the time of diagnosis was 26.6 (range 5-58) and the current average age is 34.3 years with an average follow-up period of 7.3 years (range 0-36).
Sixty percent of patients have now been living with GIST for more than five years, allowing us to generate Kaplan-Meier survival 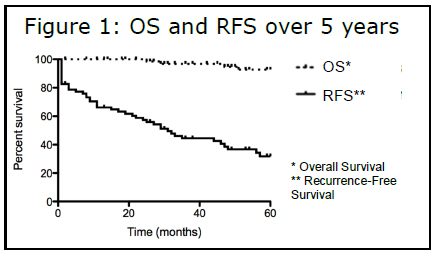 curves. Figure 1 shows the overall survival (OS) and recurrence free survival (RFS) over a five -year period. Although RFS is only 32 percent, overall survival during the same time period is quite high at 93 percent, suggesting that the vast majority of patients have an indolent form of GIST. These values did not vary significantly based on age, with a 24 percent RFS and 88 percent OS for pediatric patients versus 30 percent RFS and 93 percent OS for adult wildtype patients. For those with other types of cancer, recurrence is practically synonymous with poor prognosis. It is very striking that wildtype GIST patients are so different in this regard, in that they continue to do well despite recurrence.
curves. Figure 1 shows the overall survival (OS) and recurrence free survival (RFS) over a five -year period. Although RFS is only 32 percent, overall survival during the same time period is quite high at 93 percent, suggesting that the vast majority of patients have an indolent form of GIST. These values did not vary significantly based on age, with a 24 percent RFS and 88 percent OS for pediatric patients versus 30 percent RFS and 93 percent OS for adult wildtype patients. For those with other types of cancer, recurrence is practically synonymous with poor prognosis. It is very striking that wildtype GIST patients are so different in this regard, in that they continue to do well despite recurrence.
Additional clinical aspects that we investigated serve as important prognostic factors for assessing the risk for recurrence in KIT-mutated GIST patients. These features include the mitotic rate, size, and location of the primary tumor. We observed no significant difference in RFS in our wildtype patients based on these characteristics, suggesting that they do not predict the risk for recurrence in the wildtype setting, as they do in KIT-mutated GIST. This result is important because these characteristics are often used to make decisions about beginning adjuvant therapy. It is our recommendation that physicians do not use any of the above risk factors alone to determine the need for adjuvant therapy.
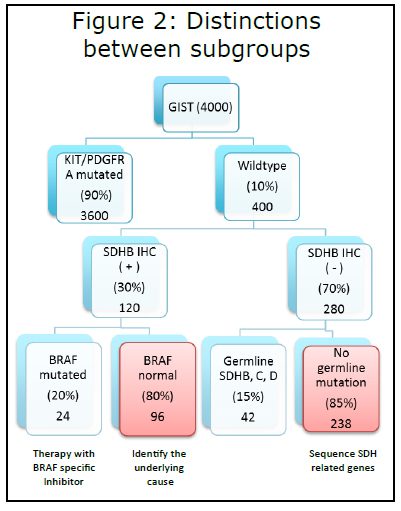
Figure 2 highlights these differences.
1) Out of the approximately 4,000 new cases of GIST diagnosed every year, only 400 are wildtype.
2) Those 400 cases can be further classified by whether the tumor is SDHB IHC positive or negative.
3) If the tumor is SDHB IHC (+), BRAF mutational testing should be performed. If a mutation is discovered, treatment using a BRAF-specific inhibitor should be initiated. We have observed that tumors with BRAF mutations are more likely to be located in the small bowel, have spindled histology, and occur more frequently in males.
4) The other 80 percent of SDHB IHC (+) GISTs that do not have a BRAF mutation will be the focus of our research initiatives to discover the underlying cause of disease. We have started germline sequencing analysis on patients with SDHB (+) GIST and their parents, to attempt to identify genes that are mutated or deleted that may play a role in tumorgenesis.
5) The majority (70%) of patients with wildtype GIST will be SDHB IHC (-). All of these patients should undergo germline testing of SDH subunits B, C, and D (also A when it becomes CLIA certified). Fifteen percent of patients will have mutations, at which point we recommend genetic screening of immediate family members. Affected family members should then undergo biochemical and radiographic surveillance.
6) Eighty-five percent of patients who are SDHB IHC (-) will not have germline mutations in sub-units B, C, and D. One hypothesis is that muta-tions in other genes im-portant in proper SDH formation and function result in the loss of SDHB staining. Thus, we are currently sequencing tumor samples for all seven recognized components of SDH to test this hypothesis.
The rationale for sub-classification is to direct treatment and to focus our research efforts to identify new therapeutic targets. Our goal is to open a treatment protocol with SDHB IHC (-) GIST, targeting the SDH pathway.
Thanks to the continued support of Life Raft Group and the patients who have participated in our clinics, we have made great progress so far. Both continue to provide essential help as we strive toward our common goal.
References:
1. Janeway KA, Kim SY, Lodish M, et al. Defects in succinate dehydrogenase in gastrointestinal stromal tumors lacking KIT and PDGFRA mutations. Proc Natl Acad Sci USA 2011;108:314-8.