Researchers have developed a test that can accurately identify over 20% of GIST patients that are unlikely to respond to currently approved treatments. In addition, it can identify which patients need a higher dose of some drugs, which patients should avoid adjuvant treatment and can even direct some patients into specific, tailored clinical trials as well as which patients should avoid specific clinical trials.
Using this test, some new GIST patients with advanced disease could completely skip treatments that have been identified as poor choices and immediately be directed into trials for their specific type of GIST, saving years of treatment with expensive drugs that often have significant side effects.
These patients can access effective treatment at a point when they are much healthier and more likely to benefit. The test uses readily available tissue blocks from a patient’s previous surgery or biopsy. Specialized, but widespread, laboratories are able to analyze the tissue to determine the likely genetic driver of almost any GIST patient’s cancer. Preliminary evidence from a large cohort of GIST patients suggests that patients having this test live longer than patients that don’t have the test; even after adjusting for multiple confounding factors.
This test will be available to all GIST patients starting in . . .
. . . 2003.
Okay, so the test isn’t brand new, but the real question is; why aren’t more people using a test that can do all of these amazing things that patients with other cancers can only dream about?
The test is called a mutational test and typically costs about a thousand dollars. This is almost always paid for by insurance. GIST experts all over the world recommend this test; and yet only about half of living patients in the LRG registry have the test. And using the LRG registry as a measure of testing is probably misleading because it represents some of the most engaged, proactive, educated patients in the world and the testing rate in the LRG registry is almost certainly higher than in the general population of GIST patients. In fact, the rate
for mutational testing in the general oncology world may be as low as 1% in the USA (in 2010) to 17% in the Netherlands (in 2012) (1,2). The testing rate for patients treated at GIST specialty centers is much higher; as high as 92% in the 2012 study from the Netherlands.(1) This is just of many reasons that GIST patients should consult with GIST expert doctors.
Researchers have made great progress in understanding GIST since 1998, and today many different sub-types of GIST are recognized. With the use of mutational testing, it’s now possible to understand the driving force for almost all GISTs. Mutational testing has been available since 2003, but its use in GIST remains shockingly low.
A standard mutational test for GIST includes testing for mutations in KIT and PDGFRA and is sufficient to find the driver mutation in about 80% to 85% of GIST patients. Additional testing, including, but not limited to, staining for SDHB is needed, but available, for those patients without a KIT or PDGFRA mutation.
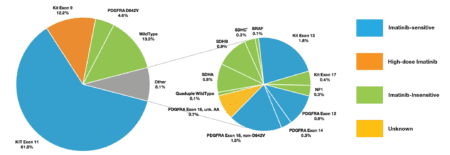
Figure 1 – Real World Evidence – Data from the LRG registry supports mutational testing
When we include all LRG patients with a known mutation type (n = 739) including the KIT exon 11 patients, two thirds (66%) are imatinib-sensitive, 12% have intermediate sensitivity, 21% are insensitive to imatinib and 1% have incomplete reporting and thus unknown sensitivity (figure 1).
Data from the LRG Registry
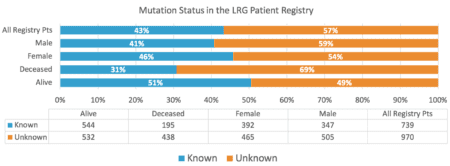
In the LRG registry, 43% of patients (n = 739) reported a mutation to the registry (known) and 57% (n = 970) reported as unknown mutation (figure 2). The unknown most likely represents that the patient did not have any testing, but there may be some cases where testing was done and the patient either did not realize that testing had been done or did not know the result. The percentage of patients with known mutation has increased over time and 67% of LRG patients diagnosed in 2013 or later reported a known mutation.
The rate is also higher if we consider only living patients, with 50.6% of living patients having a known mutation. Unfortunately, this still leaves almost half (49.4%) of the living LRG patients without testing. A very high percentage when you consider that most living LRG patients are either currently metastatic (49%) or at high risk of a recurrence (71% of patients with known risk criteria that have not had a recurrence).
Figure 2 – Mutation Status in the LRG Patient Registry
Using registry data, we calculated the overall survival times from patients with a known mutation and compared this to patients with an unknown mutation. We calculated this in
a number of different stages of disease and overall survival was longer for patients with a known mutation in every stage of treatment through 3rd line. There are a number of confounding variables that make such a comparison difficult and we have identified at least some of these variables and included them in a multivariable analysis to estimate the effects of these variables on survival (using a Cox- proportional hazards model).
Variables that affect the data
The confounding variables that we have been able to identify include:
- Date of diagnosis and date starting treatments (new treatments have improved survival over time and mutational testing rates have improved over time.).
- Age – Younger patients have increased rates of testing and longer survival due to the predominance of SDH-deficient GIST in younger patients.
- Gender – Females have longer survival times in GIST and have mutational testing at a slightly higher rate than males.
- Patients with higher risk GIST or patients undergoing adjuvant treatment are more likely to get testing.
After including these variables in the model, mutational testing remains significant. Patients with a mutational test lived longer in our analysis and the difference can be as much as 80% higher risk of death for those without testing (depending on stage).
In our opinion the most important confounding variable of all is very difficult to measure. We believe that being a proactive, engaged patient affects your survival, and proactive, engaged patients are much more likely to both ask for a mutational test and to seek out expert GIST doctors for consultations and additional treatments such as participation in clinical trials. The good news about this for patients is, that this isa variable that is almost entirely within a patients control. A patient can choose to become educated about their disease, to ask to see a GIST specialist, to ask for a mutation test, to become proactive and engaged in their care. Or if they need help, to ask a family member to be their advocate.
Mutational testing evolved over many years. With each new revelation, the evidence and value for testing became stronger and stronger. If mutational testing weren’t available and we were suddenly presented with a new test that would do all of the things that mutational testing can do today, we would proclaim a tremendous victory and it would immediately become the standard of care.
Note: Mutational testing rates are lower for GIST patients with a low risk of recurrence. These patients may be seen more often in local oncology settings and under-represented in GIST specialty centers as well as the LRG registry.
While not performing a mutational test for low risk patients not being considered for drug therapy may seem like a reasonable decision, there are other considerations, such as the risk of familial GIST for some SDH-deficient patients. Low risk patients not receiving drug therapy or mutational test should have enough testing to rule out SDH deficiency (such as staining for SDHB).
References
- Pisters, P. W. T. et al. A USA registry of gastrointestinal stromal tumor patients: changes in practice over time and differences between community and academic practices. Ann Oncol (2011). doi:10.1093/annonc/mdq773.
- Verschoor, A. J. et al. The incidence, mutational status, risk classification and referral pattern of gastro-intestinal stromal tumours in the Netherlands: a nationwide pathology registry (PALGA) study. Virchows Arch. (2018). doi:10.1007/s00428-017-2285-x.