The week of October 16 was a busy one for the Life Raft Group’s advocacy efforts. The week began with a trip to the National Organization of Rare Diseases’ 2016 Breakthrough Summit. Held annually, the Summit brings together representatives from the FDA, rare disease patient organizations, pharma, biotech and the media for two days of updates, innovation, networking and support. The LRG’s Director of Project Management, Mildred Menos, traveled to Arlington, Virginia for this year’s event to spread the word about the LRG’s newest side effect management resource – SideEQ (www.mysideeq.org) and to connect with others in the rare cancer community.
Later that week, Mildred traveled to Montreal to take part in Life Raft Group Canada’s annual GIST Day of Learning. It was Life Raft Group Canada’s largest annual meeting yet and the kinship in the room was palpable. Patients shared their stories and struggles, gave and received advice and contributed to a brainstorming session on programs and servicesLife Raft Group Canada could offer to best address the needs of the Canadian GIST community.
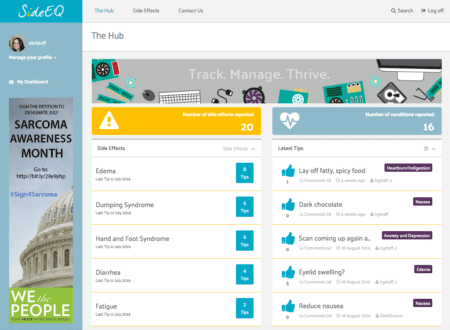
It was a busy but enlightening week as the LRG team continues to work to improve SideEQ to be as useful as possible during its beta testing period. If you would like to sign up to be a beta tester, have any feedback to contribute or would just like to sign up to receive help managing side effects please visit SideEQ at www.mysideeq.org